R/S Nomenclature
Stereoisomerism at sp3 carbon
Asymmetry in Organic Molecules
When a tetrahedral (sp3) carbon has 4 different groups attached it is able to exhibit a type of stereoisomerism that allows for 2 unique isomers which differ by their arrangment of groups within 3-dimensional space. These isomers will be non-superimposable mirrors images and are described as being enantiomers. These isomers have identical physical properties except that they rotate plane-polarized light by the same degree but in opposite directions (+/-). The enantiomeric 2-butanols below are examples of this phenomenon. Rotate the two models to see how they differ in their 3-dimensional geometry.
2-Butanols
(-)-2-Butanol
(+)-2-Butanol
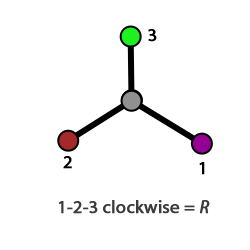
We think of enantiomers as being a left-hand/right-hand pair, and the word chiral is derived from "handedness" in Greek, so your hands are the most obvious example of being non-superimposable mirror images. Since enantiomers are isomers they need to have different names and the nomenclature system (R and S) used is based on the Cahn-Ingold-Prelog prioritization scheme. Consider the molecule CHIBrCl below; the C atom has 4 different atoms attached and so it is chiral. We order the attached groups in descending priority, firstly based on atomic mass, so here I = 1, Br = 2, Cl = 3, H = 4. We then orientate the molecule so that 4 is pointing away from us (try this with the 3-D stucture). The remaining groups are 1-2-3 clockwise, which is designated as the R stereocenter, or 1,2,3-counterclockwise, which is the S stereocenter. The rotation in the example below is clockwise so this is the R isomer of CHIBrCl.
Note: grey = C; white = H; orange = Br; green = Cl; purple = I
CHIBrCl
3-D structure
Assignment