| Chemistry
3719 - Dr.
Peter Norris Nucleophilic Substitution Reactions |
|
Nucleophilic Substitution
reactions
are transformations in which nucleophilic reagents displace a leaving
group
in an electrophilic substrate. The substrate will usually be an alcohol
(or some derivative thereof) or an alkyl halide. There are two
possible
mechanisms to be considered here, the SN1 in which the leaving
group
comes off before the nucleophile bonds (i.e. a stepwise mechanism), and
the SN2 in which the bond
making and bond breaking steps occur at the same
time (concerted mechanism). The following series of graphics may
help you visualize the events occurring during these different
mechanisms. |
|
Nucleophilic
Substitution Unimolecular
(SN1) - So far in 3719 this type of reaction most often occurs
with tertiaryalcohols which are capable of generating a tertiary
carbocation
when the leaving group is lost. We use an acid such as HCl,
HBr and HI so that water behaves as the leaving group and Cl -, Br - or
I - as the nucleophile.
Overall Reaction:
|
| Mechanism Using
t-butanol reacting with HBr
as an example, the first step of the reaction is a protonation of the
alcohol's
OH group. It is important to note that this is not the SN1 yet, it is a
very fast acid-base reaction that sets up the substitution reaction.
|
| The
actual substitution reaction begins
when the leaving group breaks away to generate a 3o cation.
This is the slowest step in the mechanism and is therefore defined as
the rate-determining
step. Since the R.D.S. only involves one species (the protonated
alcohol)
this reaction is said to be unimolecular, i.e. SN1.
|
| The
unstable carbocation (unstable since
it only has 6 electrons) is then able to react with the elecrton-rich
nucleophile
(in this case the Br - from HBr) and regain the stable octet
configuration.
This reaction is very fast (low activation barrier) and leads to the
alkyl
halide product.
|
|
Overall, the reaction results in nucleophilic
substitution of a leaving group by a nucleophile on
an electrophilic
carbon. In this case the R.D.S. is known to be unimolecular and
is therefore defined as SN1. The SN1 is classified as a stepwise
mechanism. The following animation
puts the mechanistic
events in sequence:
|
|
Nucleophilic Substitution
Bimolecular
(SN2) - So far in 3719 this
type of reaction most often occurs with primary alcohols which are
not capable of generating a decent carbocation when the leaving group
is
lost, instead the bond making and bond breaking events occur
simultanously
(i.e. a concerted pathway). We use an acid such as HCl, HBr and
HI so that water behaves as the leaving group and Cl -, Br - or I - as
the nucleophile. Overall
Reaction
|
|
Mechanism
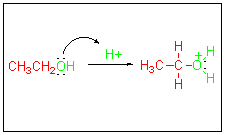
Using
ethanol and HBr as a typical
example,
the first step in the alcohol to alkyl halide conversion is an
acid-base
reaction to form a good leaving group (H2O). Notice again
that
this step is a prelude to the substitution reaction.
|
|
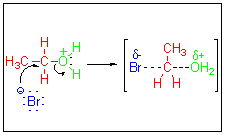
The sustitution step occurs when
the
nucleophile
(Br - in this case) begins to bond to the electrophilic carbon at
the
same time that the leaving group (in this case H2O)
begins
to break away. The system passes through a transition state which is
depicted
using partial bonds. Note there are no intermediates such as
carbocations
formed here. This step is rate determining and is bimolecular,
therefore
SN2.
|
|
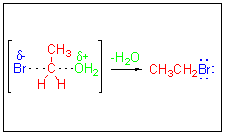
The complete breaking of the C-O bond and the complete formation of the C-Br bond leads to the alkyl halide product. Overall,
the reaction results in nucleophilic
substitution of a leaving group by a nucleophile on
an
electrophilic
carbon. In this case the R.D.S. is known to be bimolecular and
is
therefore defined as SN2. The SN2 is classified as a concerted
mechanism.
|
| The
following animation puts the
mechanistic
events in sequence:
|
| Most alcohol substrates have the option of either SN2 or SN1 and the path taken depends on structure. At this point tertiary alcohols will follow the SN1 pathway (decent carbocation, stepwise mechanism), and primary alcohols will follow the alternate SN2 pathway (concerted mechanism). |
| Peter
Norris, Ph.D. |